|
|
Post by SeaRat on Oct 29, 2016 20:45:02 GMT -8
This thread is being created for those who wish to see the ways that the two types of depth gauges were modified for use in fresh water and at altitude. In addition, I have enclosed the Cross Tables for "Theoretical Depth At Altitude for Given Actual Depths." This information comes from a conference I attended on altitude diving for the USAF in about 1975. 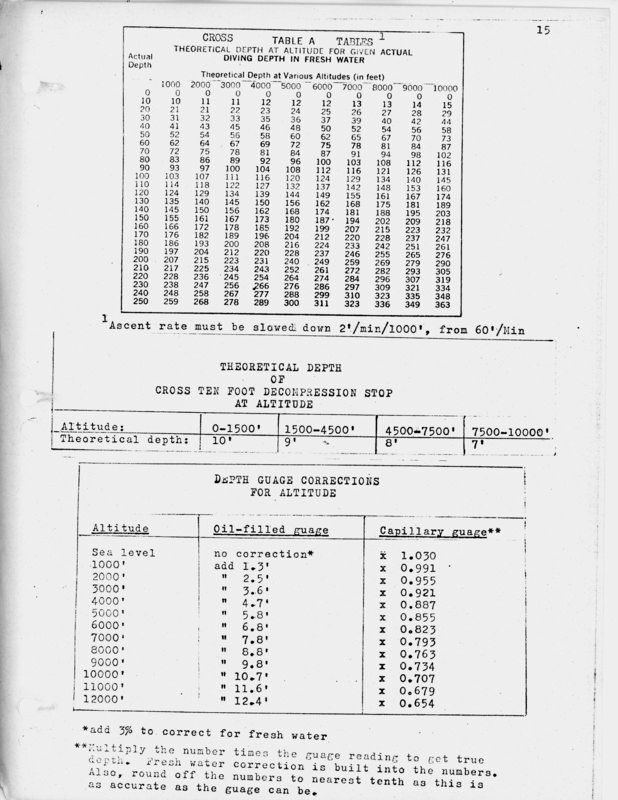 You will notice that there are three tables. They are: --Cross Tables, "Theoretical Depth at Altitude for Given Actual Diving Depth in Fresh Water"--This table was developed by E.R. Cross to show the theoretical depth that should be used with the U.S. Navy Diving Tables for the actual depth (line measured depth) in fresh water. For instance, if I were to dive in Clear Lake, Oregon at a depth of 4500 feet, I would use the 5000 foot column at 100 feet, and that is the depth I would use for the U.S. Navy Diving Tables that are used at sea level. On this depth, I would have to use 120 feet in the U.S. Navy Diving Tables to get the correct decompression times for that altitude. --"Theoretical Depth of Cross Ten Foot Decompression Stop at Altitude"-- For the above dive, I would need to modify the depths and at 5000 feet, the ten foot stop would be at 8 feet actual depth (see below for depth gauge corrections). --"Depth Gauge Corrections for Altitude"--Depending upon the type of gauge you use, the depths at altitude will be inaccurate, but in different ways. This is critical in determining the depth of the dive, and the depth of decompression and/or safety stops. Fer instance, for that dive in Clear Lake at 4500 feet, you would go to the 5000 foot correction for a bourdon tube ("oil-filled") gauge. For this gauge, if my depth gauge read 60 feet, I would need to add 5.8 feet to that reading, which would make it a dive to 66 feet, and I would use the 70 foot no-decompression table, or use 70 feet on the decompression tables for sea level. If my gauge read 60 feet and it was a water-filled capillary gauge (the ones where the water column fills a small tube, and that tube is calibrated for different depths, I would have to multiply it by a number to get the actual depth. Capillary gauges read deep at altitude, and so if you use the capillary gauge depth, that would be a compensation for the altitude. But to get actual depth in Clear Lake, at about 5000 feet altitude, you would need to multiply the capillary gauge reading by the number, and at 5000 feet that number is 0.885. So a 60 feet depth reading would then be: 60 x 0.885 = 53 feet actual depth I hope this little thread helps with those who are using vintage gear in fresh water at altitude. John |
|
|
|
Post by cstmwrks on Oct 30, 2016 7:31:46 GMT -8
|
|
|
|
Post by cnotthoff on Oct 30, 2016 9:13:37 GMT -8
The beauty of a capillary depth gauge is that it is altitude adjusted and reads in fsw no matter what fluid your diving in. At the surface, the tube is full of atmospheric-pressure air even at altitude. When you have doubled that pressure at whatever depth, a capillary gauge will read 33 ft. This is perfect for entering into dive tables that are designed to work for dives in sea water. The drawback of a capillary gauge is that is really hard to read precisely at deeper depths.
If you create a capillary gauge that reads your actual depth in fresh water, you then need to correct to fsw to use dive tables. All depth gauges are really pressure gauges. For decompression considerations, your actual depth is not really important. What is important is the change in pressure relative to atmospheric pressure at the surface. Bourdon-tube gauge readings do require corrections to enter most dive tables.
One of the most important things to consider when diving at altitude is that a diver traveling from sea level will have residual nitrogen from being saturated at that higher atmospheric pressure. Cross covers this in his book
Good Dives,
Charlie
|
|
|
|
Post by cstmwrks on Oct 30, 2016 11:15:45 GMT -8
The beauty of a capillary depth gauge is that it is altitude adjusted and reads in fsw no matter what fluid your diving in. At the surface, the tube is full of atmospheric-pressure air even at altitude. When you have doubled that pressure at whatever depth, a capillary gauge will read 33 ft. This is perfect for entering into dive tables that are designed to work for dives in sea water. The drawback of a capillary gauge is that is really hard to read precisely at deeper depths. If you create a capillary gauge that reads your actual depth in fresh water, you then need to correct to fsw to use dive tables. All depth gauges are really pressure gauges. For decompression considerations, your actual depth is not really important. What is important is the change in pressure relative to atmospheric pressure at the surface. Bourdon-tube gauge readings do require corrections to enter most dive tables. One of the most important things to consider when diving at altitude is that a diver traveling from sea level will have residual nitrogen from being saturated at that higher atmospheric pressure. Cross covers this in his book Good Dives, Charlie The ffw capillary was just one part of the overall project. I wanted to make a converted table / gauge set to remove as much on site conversions of information as possible. Not so much for decompression but more of a square profile repeat shallow water dive table. Years ago I had made my own crude dive planner work sheet for high altitude 02 diving. It was simple and based on the duration limits of the gear I was using at the time. Your pointing out that a depth gauge is just a pressure gauge calibrated to read in feet just got me thinking about a gauge / table set that works and reads in pressure only. Essentially a universal table that works in fresh / sea / altitude as the gauge reads max pressure attained and the table calculations work on time exposure at that pressure level. Right now it's all in the "thinking about it" stage. I have not done any real number crunching on this project. |
|
|
|
Post by SeaRat on Oct 30, 2016 16:44:56 GMT -8
Cstmwrks,
There are a couple of references I have about making your own capillary depth gauge. They are pretty easy, but don't have the tubing in a straight line like your example does. They use a plastic rectangle, and have it taking several turns to make a doubled rectangle, with depth markings on the outside. But you could use both sides, and have one calibrated for salt water, and one for fresh water. But none of them are for altitude diving. You would need to use the calculation, or give one on another set, for the altitude you are diving. Realize that if you use the numbers on the capillary gauge, you will automatically adjust for altitude, as Charlie is stating above.
John
|
|
|
|
Post by cstmwrks on Oct 30, 2016 18:53:17 GMT -8
Cstmwrks, have one calibrated for salt water, and one for fresh water. But none of them are for altitude diving. You would need to use the calculation, or give one on another set, for the altitude you are diving. Realize that if you use the numbers on the capillary gauge, you will automatically adjust for altitude, as Charlie is stating above. John Hi John, Do you mean that the capillary gauge adjusts for altitude by virtue of the fact it is nothing more than an open ended tube? However the reading it gives in feet will be off as the gauge is calibrated at sea level pressure? Like I said to Charlie, I've only been thinking about this. No hard math yet. Also mentioned that a simple and universal table set could be based on pressure readings. Not depth. All tables and computers run there math on time and pressure. Then it's converted to feet for our simple minds to understand. If you cut out thinking in terms of feet and used a pressure gauge reading and a pressure based dive table set it would work in fresh water, sea water and altitude. |
|
|
|
Post by SeaRat on Oct 31, 2016 13:12:34 GMT -8
Cstmwrks,
Capillary Gauge
Here is what I mean. If you imagine the capillary gauge as an absolute pressure gauge, you would have the weight of the atmosphere (atm) on top of the weight of the water. But it is showing zero at sea level, as it is out of water until you put the gauge underwater. As you go down in depth, the pressure builds, until at 33 feet (10 meters) you are at twice the surface pressure, or 2 atm absolute pressure. The water will have traveled up the column half way. At 66 feet (20 meters) you are at 3 atm absolute, and the water will have traveled up the column 2/3 of the way (leaving only 1/3 of the original amount of air. From this you can get an idea of the relationship, and this is at sea level.
Now, let's say you go to altitude. There, the absolute pressure is not 1 atm at the surface. It is some percentage of one atm, depending upon how high you are in the air column. But as you descend, the water still weighs as much as it did at sea level (assuming fresh water, not salt water). So the pressure of the water column will remain the same, but it will start with less air density. This will cause the capillary gauge to read deep.
For example, if you dive Lake Tahoe (I did that once, a long time ago, for the Altitude Diving Conference was at Lake Tahoe), you are diving at an altitude of 6,224 feet. We'll round that off to 6000 feet. Using the table above, the correction for a capillary depth gauge is to multiply the depth reading times 0.823. So if I dive to 80 feet on the capillary gauge, this must be modified to get a true depth reading:
80 ft x 0.823 = 65.84 ft = 66 ft
So our true depth is about 66 feet, rather than 80 feet. If I use the 80 feet on the depth gauge with the U.S. Navy Diving Tables, than I am being conservative, as I was not to that depth.
Now, another way that the capillary depth gauge will read deep is that it uses the air, usually at surface temperature. But lake waters, especially Lake Tahoe at depth, is much colder. So that will also cause the capillary gauge air to contract, resulting in an additional error in depth, reading deeper.
So there are two factors happening with the capillary gauge, altitude and temperature, which will cause it to read deeper, and give you a "conservative" reading for decompression purposes.
Oil-Filled Bourdon Tube Depth Gauge
Now let's look at the oil-filled Bourdon tube depth gauge. Here, the gauge will read shallow. For the same dive in Lake Tahoe, we need to add 6.8 feet to the reading. So the dive where the gauge is reading 80 feet will actually be to:
80 ft + 6.8 ft = 86.8 ft = 87 ft
Therefore, we would need to use 90 feet when determining no-decompresssion limits or decompression stops. This is not "conservative," and could lead to a diver who doesn't know the difference to use a shallower decompression schedule than the actual depth needed for this dive. On top of that, because it is in fresh water, we need to add an additional 3% to this reading.
87 ft x 0.03 = 2.61 ft = 3 ft
So in addition to the 87 feet, we add 3 more feet to get 90 feet of actual depth for this dive, that was reading originally on this type of gauge 80 feet.
Pressure Readings
From the U.S. Navy Diving Manual, March 1970:
The U.S. Navy Diving Manual goes on to say that fresh water weighs 62.4 pounds per cubic foot. Sea water therefore exerts 1.026 times more pressure than fresh water, and thus the corrections for depth for a Bourdon tube depth gauge.
Since there is this difference, we need to do a slightly different calculation.
33 ft/atm salt water x 62.4 pounds/ft3 of fresh water = 2059.2 pounds / square foot.
2059.2 pounds per square foot / 144 square inches per square foot = 14.3 pounds / square inch. But an atmosphere is 14.7 psi, so we need to divide that by 14.3 psi to get a conversion factor for fresh water.
14.7 psi / 14.3 psi = 1.02797 = 1.028
1.028 is the correction factor for converting the pressure to one atmosphere in fresh water.
33 ft/atm x 1.02797 = 33.92 ft/atm fresh water, = 34 ft/atm fresh water
So to make the capillary gauge specifically for fresh water, the half-way point of the water column should be marked at 34 feet, and not 33 feet.
By the way, this is so much easier in the metric system. I may do this via metric later, if I find the time.
John
|
|
|
|
Post by nikeajax on Oct 31, 2016 14:10:00 GMT -8
 Uhm, hey Mark, did'ja get all that; I heard ol' man Ratliff is givin' us a test at the end of the class t'morrow... Uhm, hey Mark, did'ja get all that; I heard ol' man Ratliff is givin' us a test at the end of the class t'morrow...
|
|
|
|
Post by cstmwrks on Oct 31, 2016 16:58:20 GMT -8
Cstmwrks, By the way, this is so much easier in the metric system. I may do this via metric later, if I find the time. John Hi John, OK the long version is easy for me to understand. When you made that short response I truly could not make heads or tails of what you attempted to say. I clipped off everything but your comment on the metric system. Much as I hate the system if for no other reason than my brain is conditioned to think in feet and psi, it is more simple just because everything is in units of 10. And every 10 meters = 1 ATM 0r 1 BAR if we want to be super picky on a couple of kilopascals. Anyhow I keep thinking about avoiding as much conversion of info as possible. Lending more to not thinking in terms of feet but in absolute pressure. Oddly enough that pretty well describes the metric system. |
|






 Uhm, hey Mark, did'ja get all that; I heard ol' man Ratliff is givin' us a test at the end of the class t'morrow...
Uhm, hey Mark, did'ja get all that; I heard ol' man Ratliff is givin' us a test at the end of the class t'morrow...
